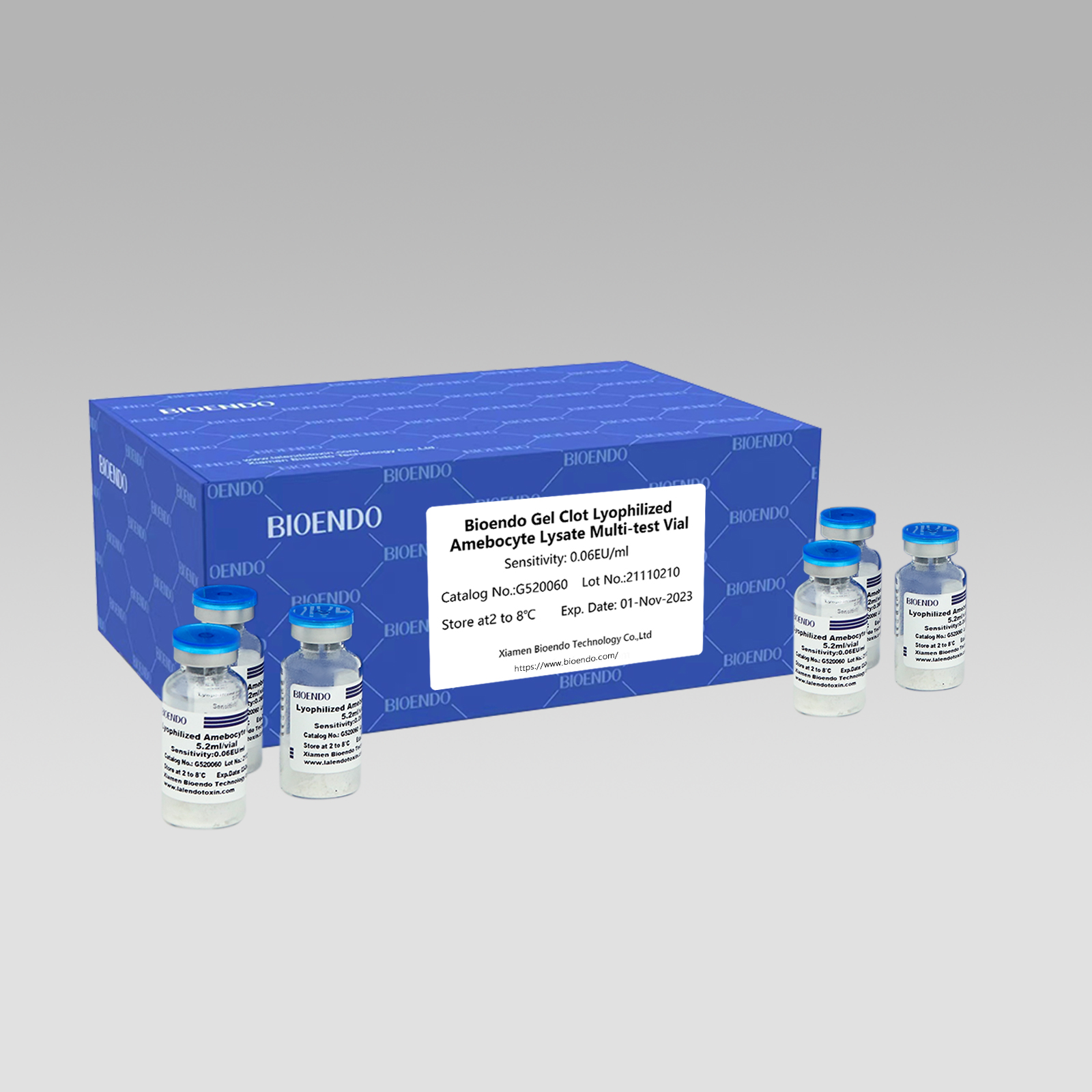
IGel Clot Lyophilized Amebocyte Lysate Multi-test Vial G52
Uthotho lwe-Bioendo G52 lusetyenziswa ikakhulu kumsebenzi wokulingauvavanyo lwebhaktiriya endotoxinnjengenkqubo yeBioassay.
1. Ulwazi lweMveliso
Indlela yeGel Clot I-Lyophilized Amebocyte Lysate I-Multi-test Vial yi-Lyophilized Amebocyte Lysate reagent ekhetha kwaye isebenzise ubuchule behlwili yejeli ukubona i-endotoxin okanye ipyrogen.
Njengendlela ebanzi, uvavanyo lwe-gel-clot lwe-endotoxin lulula kwaye alufuni isixhobo esikhethekileyo nesibizayo.I-Bioendo ibonelela nge-Gel Clot I-Lyophilized Amebocyte Lysate - i-reagent ye-LAL kwi-5.2ml nge-vial nganye.
2. IiParameters zeMveliso
Uluhlu lobuthathaka: 0.03EU/ml, 0.06EU/ml, 0.125EU/ml, 0.25EU/ml, 0.5 EU/ml
3. Isicelo seMveliso
I-endotoxin yemveliso yokugqibela (pyrogen) isiqinisekiso, amanzi okuhlambauvavanyo lwe-endotoxin, Impahla engekasilwauvavanyo lwe-endotoxinokanye ukubeka iliso kwinqanaba le-endotoxin ngexesha lenkqubo yokuvelisa iinkampani zamachiza okanye abavelisi bezixhobo zonyango.
Phawula:
I-Lyophilized Amebocyte Lysate (i-LAL reagent) eyenziwa yi-Bioendo yenziwe nge-lysate ye-amebocytes (iiseli zegazi ezimhlophe) ukusuka ku-crab we-horseshoe.
Le reagent ekhethekileyo ibe sisixhobo esibalulekileyo kumashishini amayeza kunye nezixhobo zonyango ukuze kubonwe i-endotoxins yebhaktheriya.I-amebocyte ye-horseshoe crab iqulethe into ebizwa ngokuba yi-Lyophilized Amebocyte Lysate, esabela kwi-endotoxins yebhaktiriya ngokwenza ihlwili efana nejeli.Oku kusabela sisiseko sovavanyo lwe-LAL, olusetyenziselwa ukuqinisekisa ukhuseleko lwezixhobo zonyango, amachiza, kunye nezinye iimveliso ezidibana nomzimba womntu.
Ukusetyenziswa kwe-reagent ye-LAL kuye kwayiguqula inkqubo yeukufunyanwa kwe-endotoxinkwinkalo yezonyango kunovavanyo loMvundla.Ubuntununtunu bayo obungalinganiyo kunye nobuchwephesha buyenza ibe licandelo elibalulekileyo kulawulo lomgangatho kunye noqinisekiso lokhuseleko lwamayeza, i-biologics, kunye nezixhobo zonyango.Uvavanyo lwe-LAL yindlela ekhawulezayo nethembekileyoukufunyanwa kwe-endotoxin, ibonelela ngeziphumo kwimizuzu engama-60.Oku kusebenza kakuhle kuvumela izigqibo ezikhawulezayo nezichanekileyo malunga nokukhululwa kweemveliso, ekugqibeleni kuphuculwe ukhuseleko jikelele kunye nokusebenza kakuhle kwezonyango zonyango kunye nezixhobo.
I-Bioendo's Lyophilized Amebocyte Lysate (i-LAL reagent) iveliswa phantsi kwemigangatho engqongqo yomgangatho wokuqinisekisa ukusebenza kwayo kunye nokuthembeka.Inkampani izinikele ekusebenziseni iindlela ezizinzileyo ekuvuneni oononkala bamahashe ukunciphisa naziphi na iimpembelelo ezimbi kubemi babo.Ngokubeka phambili intlalontle yezi zidalwa, i-Bioendo iqinisekisa ukubonelelwa okuqhubekayo kwesi sixhobo sixabisekileyo sokwenziwa kwee-reagents ze-LAL.Ukongeza, iinzame zophando oluqhubekayo kunye nophuhliso zijolise ekuphuculeni ukusebenza kunye nobuninzi beI-LAL yokuvavanya i-endotoxin, ukuqhubela phambili usetyenziso lwazo kumashishini ezonyango kunye namayeza.
Indlela yehlwili yeGelUvavanyo lwe-LAL, i-reagent ehlaziyiweyo ye-lysate ifumana ubuncinane iimvavanyo ze-50 nge-vial nganye:
| Inombolo Yekhathalogu | Uvakalelo (EU/ml okanye IU/ml) | ml/ivial | Iimvavanyo / Vial | Iingqayi / Pack |
| G520030 | 0.03 | 5.2 | 50 | 10 |
| G520060 | 0.06 | 5.2 | 50 | 10 |
| G520125 | 0.125 | 5.2 | 50 | 10 |
| G520250 | 0.25 | 5.2 | 50 | 10 |
| G520500 | 0.5 | 5.2 | 50 | 10 |
Imeko yemveliso:
I-Lyophilized Amebocyte Lysate - i-LAL ye-reagent sensitivity kunye ne-Control Standard Endotoxin potency ivavanywa ngokuchasene ne-USP ye-Reference Standard Endotoxin.Iikiti ze-reagent ye-Lyophilized Amebocyte ziza nomyalelo wemveliso, iSatifikethi sokuHlalutya, i-MSDS.
Yintoni umahluko phakathi kwe-Bioendo yovavanyo lwe-vial enye kunye ne-multiple test vial?
● Uvavanyo olunye: phinda wenze olunyeuvavanyo lwe-limulus lysateokanye ufowunelwelimulus amebocytengamanzi BET kwivial yeglasi okanye i-ampoule yeglasi.
● Uvavanyo oluninzi: hlanganisa kwakhona i-reagent ye-lysate ngamanzi e-BET, uze udibanise inani eliphawulweyo le-lysate reagent elandela i-COA kwityhubhu yokusabela okanye ipleyiti yokusetyenziswa.Akukho mahluko kwinkqubo yesampulu yangaphambili yokucubungula;ngokobungakanani bovavanyo olusetyenzisiweyo, ubungakanani besampulu esetyenziselwa uvavanyo olunye bukhulu kunobungakanani besampulu esetyenziselwa iimvavanyo ezininzi.
Kutheni ikhithi yokuvavanya ihlwili yejeli ye-G52 ikhethekileyo yobuninzi beesampulu zobuninzi?
1. Uvavanyo oluninzi lwe-reagent ye-LAL yokufunyaniswa kwe-endotoxin kwizicelo zeesampulu zobunzima 'iinkqubo zokuvavanywa kwe-LAL.
2. Uluhlu lwe-G52 lwe-Gel clot endotoxin assay multi test vial vial akukho mfuneko yomfundi we-microplate ephucukileyo.Kuvavanyo lwe-LAL inkqubo yayo yokufukamela ngokuhlamba ngamanzi okanye nge-incubator eyomileyo yobushushu sisixhobo esiluncedo.
3. Umgangatho ophezulu wokuphela kwe-endotoxin ityhubhu yamahhala (<0.005EU / ml) kunye nomgangatho ophezulu weengcebiso zamahhala ze-pyrogen (<0.005EU / ml) njengento yokusetyenziswa okuqinisekisiweyo ukuqinisekisa umphumo ochanekileyo.
4. Ukukhetha i-bioendo eyodwa yovavanyo lwe-LAL okanye i-vial yovavanyo emininzi ye-LAL ngobuninzi beesampuli, ekujoliswe kuyoUvavanyo lwe-LAL lwepyrogensukubhaqwa.
Iimveliso ezinxulumeneyo kuvavanyo lovavanyo lwe-endotoxin:
Amanzi oVavanyo lwe-Endotoxins ye-Bacterial (BET), Cebisa i-TRW50 okanye i-TRW100
I-Endotoxin yasimahla ityhubhu yeglasi (ityhubhu yokuxutywa), Ncoma i-T1310018 kunye ne-T107540
Iingcebiso zamahhala zePyrogen, Ncoma i-PT25096 okanye i-PT100096
Pipettor, Ncoma iPSB0220
Uvavanyo lweTube Rack
I-Incubation Instrument (iBhafu yamanzi okanye i-Dry Heat Incubator), ukucebisa i-Bioendo Dry Heat Incubator TAL-M2 yimingxuma engama-60 enye imodyuli.
Umxube weVortex, Ncoma iVXH.
Ukulawula i-Endotoxin esemgangathweni, i-CSE10V.